“Designer” protein-modifying enzymes for biomedicine, biotechnology and synthetic biology
Enzymes that catalyze site-specific protein modifications, including kinases, methyltransferases, proteases and protein ligases, among others, play vital roles in regulating cellular processes. Understanding the substrate specificity of these enzymes is instrumental to gain insight into their physiological mechanisms, establish potential druggable targets, and leverage or redesign their specificity. Successes along this axis would enable a variety of applications in enzyme therapeutics, biorthogonal chemistry, mass spectrometry and synthetic biology. In the Denard lab, we utilize and establish methods of protein engineering and design to redefine and redesign the substrate specificity of protein-modifying enzymes in order to repurpose them as novel therapeutic and diagnostic modalities. We expect that novel protein and cell-based therapeutic modalities will expand our toolset of biologics for management of chronic illnesses, inflammations and cancer. Our research is multi-disciplinary, combines Principles of Synthetic Biology, High-throughput Screening, Next-Generation Sequencing, Bioinformatics and Structural Modeling.
Catalytic modulation of disease-associated proteins
|
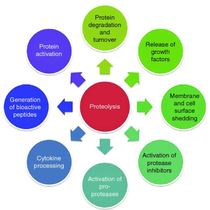
In one area of focus, we aim to evolve the specificity of proteases to target misfolded and aberrant proteins involved in neurodegenerative, autoimmune diseases and cancer. We hypothesize that catalytic degradation of disease-related proteins can fight diseases in ways that can be complementary to and mechanistically distinct from current therapeutic approaches. Unlike antibodies and small molecules, proteases can catalytically degrade their targets, making them attractive therapeutic agents that would enable novel strategies to target diseases. Protease substrate specificity engineering represents a major step in the design of therapeutic proteases.
|
High-throughput discovery and engineering of protease modulatory proteins
|
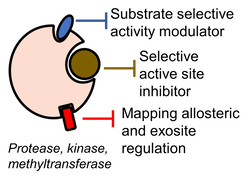
An important question in drug discovery is whether one can reprogram the substrate preference and activity of an enzyme rather than completely inhibiting its catalytic activity. Substrate-selective modulation could represent a paradigm shift in how we target diseases such as diabetes, Alzheimer’s and heart disease. The Denard Lab is developing high-throughput functional screens and statistical machine learning approaches to concurrently map enzyme distal regulation, discover and ultimately predict properties of ligand that elicit, induce, and stabilize substrate-selective conformations.
|
Discovering, profiling, and engineering enzyme-catalyzed protein ligations and amino acid side chain modifications
The ability to introduce a site-specific, regio- and chemoselective modification into a protein, peptide, or biomaterial, is at the heart of most chemical biology tools. Many of the enzymes that catalyze these transformations have become mainstay in the chemical biology toolbox, including sortases, protein ligases, and several enzymes that modify specific amino acid side chains. Yet, most of these enzymes are restricted by their narrow native sequences, which limits their general applicability. The Denard lab takes a holistic approach to substrate specificity engineering, combining bioinformatics, genome mining, high-throughput screening, next-gen sequencing, computational modeling, and machine learning to 1) discover and characterize enzymes with novel substrate specificities, 2) discover previously unknown substrate profiles in known enzymes, and 3) engineer new substrate recognition. We are applying this pipeline to sortases, plant protein ligases, and formylglycine-generating enzymes.